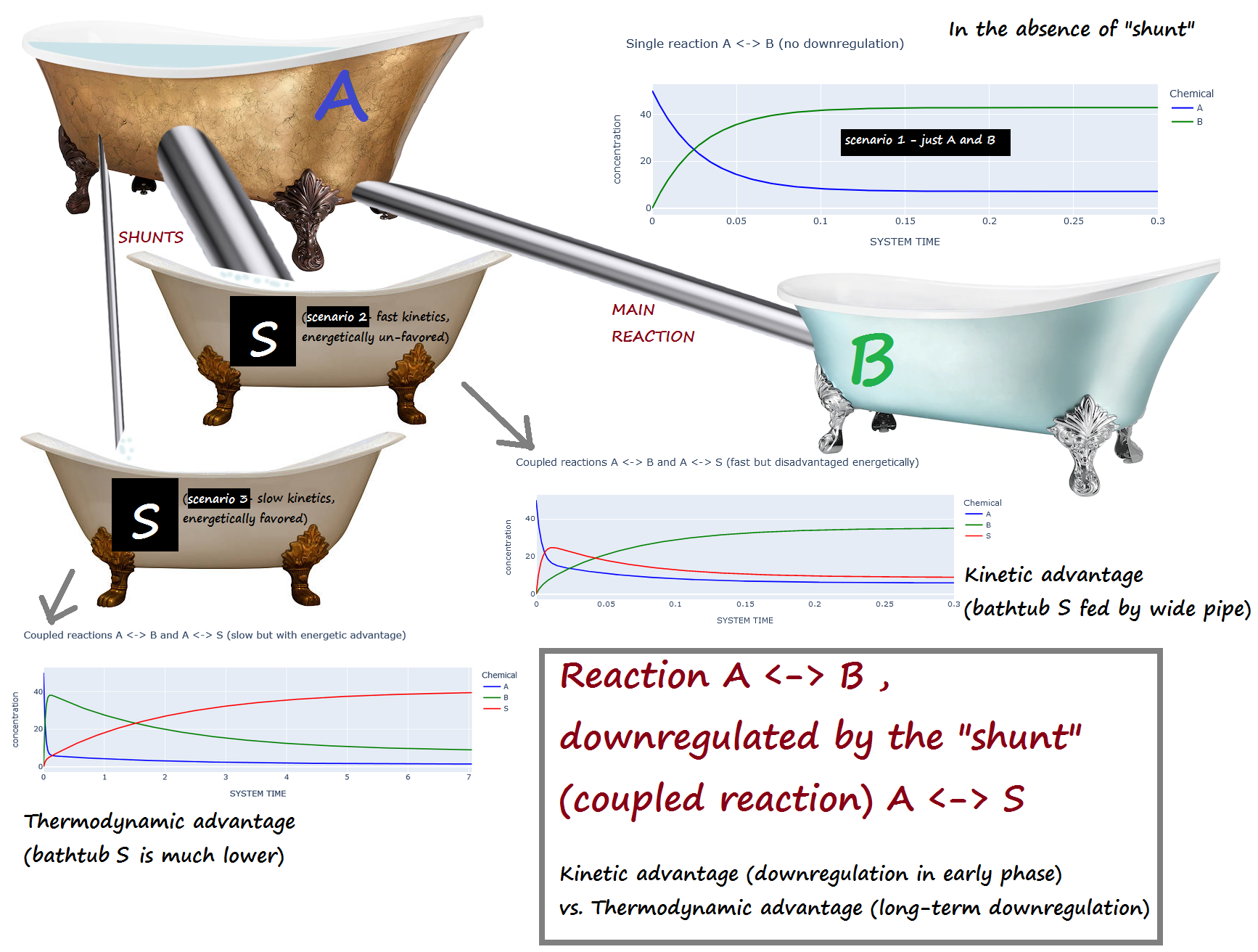
A <-> B , downregulated by the "shunt" (coupled reaction) A <-> S¶
Kinetic advantage (downregulation in early phase) vs. Thermodynamic advantage (long-term downregulation)¶
Scenario 1 : No downregulation on A <-> B
Scenario 2 : The shunt (A <-> S) has a kinetic advantage but thermodynamic DIS-advantage compared to A <-> B
(i.e. A <-> S is fast, but energetically unfavored)
Scenario 3 : The shunt (A <-> S) is has a kinetic DIS-advantage but a thermodynamic advantage compared to A <-> B
(i.e. A <-> S is slow, but energetically favored)
All reactions are 1st order, mostly forward. Taken to equilibrium.
LAST REVISED: June 23, 2024 (using v. 1.0 beta36)
Bathtub analogy:¶
A is initially full, while B and S are empty.
If the "shunt" S is present, scenario 2 corresponds to a large pipe and a small elevation change...
while scenario 3 corresponds to a narrow pipe and a large elevation change.
import set_path # Importing this module will add the project's home directory to sys.path
Added 'D:\Docs\- MY CODE\BioSimulations\life123-Win7' to sys.path
from experiments.get_notebook_info import get_notebook_basename
from life123 import ChemData as chem
from life123 import UniformCompartment
import plotly.express as px
from life123 import GraphicLog
# Initialize the HTML logging (for the graphics)
log_file = get_notebook_basename() + ".log.htm" # Use the notebook base filename for the log file
# Set up the use of some specified graphic (Vue) components
GraphicLog.config(filename=log_file,
components=["vue_cytoscape_2"],
extra_js="https://cdnjs.cloudflare.com/ajax/libs/cytoscape/3.21.2/cytoscape.umd.js")
-> Output will be LOGGED into the file 'down_regulate_1.log.htm'
Scenario 1: A <-> B in the absence of the 2nd reaction¶
Initialize the System¶
Specify the chemicals and the reaction
# Specify the chemicals
chem_data = chem(names=["A", "B"])
# Reaction A <-> B
chem_data.add_reaction(reactants=["A"], products=["B"],
forward_rate=30., reverse_rate=5.)
chem_data.describe_reactions()
Number of reactions: 1 (at temp. 25 C)
0: A <-> B (kF = 30 / kR = 5 / delta_G = -4,441.7 / K = 6) | 1st order in all reactants & products
Set of chemicals involved in the above reactions: {'A', 'B'}
Set the initial concentrations of all the chemicals¶
dynamics = UniformCompartment(chem_data=chem_data, preset="fast")
dynamics.set_conc(conc={"A": 50.}, snapshot=True)
dynamics.describe_state()
SYSTEM STATE at Time t = 0:
2 species:
Species 0 (A). Conc: 50.0
Species 1 (B). Conc: 0.0
Set of chemicals involved in reactions: {'A', 'B'}
Run the reaction¶
dynamics.set_diagnostics() # To save diagnostic information about the call to single_compartment_react()
# The changes of concentrations vary very rapidly early on; automated variable timesteps will take care of that
dynamics.single_compartment_react(initial_step=0.001, duration=0.3,
snapshots={"initial_caption": "1st reaction step",
"final_caption": "last reaction step"},
variable_steps=True)
37 total step(s) taken
Number of step re-do's because of negative concentrations: 0
Number of step re-do's because of elective soft aborts: 0
Norm usage: {'norm_A': 22, 'norm_B': 17, 'norm_C': 16, 'norm_D': 16}
dynamics.get_history()
| SYSTEM TIME | A | B | caption | |
|---|---|---|---|---|
| 0 | 0.000000 | 50.000000 | 0.000000 | Initialized state |
| 1 | 0.001000 | 48.500000 | 1.500000 | 1st reaction step |
| 2 | 0.002000 | 47.052500 | 2.947500 | |
| 3 | 0.002800 | 45.935030 | 4.064970 | |
| 4 | 0.003600 | 44.848849 | 5.151151 | |
| 5 | 0.004400 | 43.793081 | 6.206919 | |
| 6 | 0.005200 | 42.766875 | 7.233125 | |
| 7 | 0.006000 | 41.769403 | 8.230597 | |
| 8 | 0.007200 | 40.315088 | 9.684912 | |
| 9 | 0.008400 | 38.921854 | 11.078146 | |
| 10 | 0.009600 | 37.587136 | 12.412864 | |
| 11 | 0.010800 | 36.308476 | 13.691524 | |
| 12 | 0.012000 | 35.083520 | 14.916480 | |
| 13 | 0.013800 | 33.323259 | 16.676741 | |
| 14 | 0.015240 | 32.003766 | 17.996234 | |
| 15 | 0.016680 | 30.750777 | 19.249223 | |
| 16 | 0.018840 | 28.966018 | 21.033982 | |
| 17 | 0.020568 | 27.646153 | 22.353847 | |
| 18 | 0.022296 | 26.406114 | 23.593886 | |
| 19 | 0.024888 | 24.658551 | 25.341449 | |
| 20 | 0.026962 | 23.387332 | 26.612668 | |
| 21 | 0.029035 | 22.208373 | 27.791627 | |
| 22 | 0.032146 | 20.568281 | 29.431719 | |
| 23 | 0.034634 | 19.399045 | 30.600955 | |
| 24 | 0.038366 | 17.797935 | 32.202065 | |
| 25 | 0.041352 | 16.684379 | 33.315621 | |
| 26 | 0.045831 | 15.188610 | 34.811390 | |
| 27 | 0.050310 | 13.927325 | 36.072675 | |
| 28 | 0.057029 | 12.331983 | 37.668017 | |
| 29 | 0.062404 | 11.355820 | 38.644180 | |
| 30 | 0.070466 | 10.167025 | 39.832975 | |
| 31 | 0.082559 | 8.887006 | 41.112994 | |
| 32 | 0.094652 | 8.148772 | 41.851228 | |
| 33 | 0.112792 | 7.510122 | 42.489878 | |
| 34 | 0.140002 | 7.160360 | 42.839640 | |
| 35 | 0.180816 | 7.135357 | 42.864643 | |
| 36 | 0.242039 | 7.151428 | 42.848572 | |
| 37 | 0.333872 | 7.123879 | 42.876121 | last reaction step |
dynamics.explain_time_advance()
From time 0 to 0.002, in 2 steps of 0.001 From time 0.002 to 0.006, in 5 steps of 0.0008 From time 0.006 to 0.012, in 5 steps of 0.0012 From time 0.012 to 0.0138, in 1 step of 0.0018 From time 0.0138 to 0.01668, in 2 steps of 0.00144 From time 0.01668 to 0.01884, in 1 step of 0.00216 From time 0.01884 to 0.0223, in 2 steps of 0.00173 From time 0.0223 to 0.02489, in 1 step of 0.00259 From time 0.02489 to 0.02904, in 2 steps of 0.00207 From time 0.02904 to 0.03215, in 1 step of 0.00311 From time 0.03215 to 0.03463, in 1 step of 0.00249 From time 0.03463 to 0.03837, in 1 step of 0.00373 From time 0.03837 to 0.04135, in 1 step of 0.00299 From time 0.04135 to 0.05031, in 2 steps of 0.00448 From time 0.05031 to 0.05703, in 1 step of 0.00672 From time 0.05703 to 0.0624, in 1 step of 0.00537 From time 0.0624 to 0.07047, in 1 step of 0.00806 From time 0.07047 to 0.09465, in 2 steps of 0.0121 From time 0.09465 to 0.1128, in 1 step of 0.0181 From time 0.1128 to 0.14, in 1 step of 0.0272 From time 0.14 to 0.1808, in 1 step of 0.0408 From time 0.1808 to 0.242, in 1 step of 0.0612 From time 0.242 to 0.3339, in 1 step of 0.0918 (37 steps total)
dynamics.plot_history(colors=["darkturquoise", "green"], title="Single reaction A <-> B (no downregulation)",
show_intervals=True)
Notice the intersection at the exact midpoint of the 2 initial concentrations (50 and 0):¶
dynamics.curve_intersect('A', 'B', t_start=0, t_end=0.1)
(0.024381560069949713, 25.0)
# Verify that all the reactions have reached equilibrium
dynamics.is_in_equilibrium()
0: A <-> B
Final concentrations: [A] = 7.124 ; [B] = 42.88
1. Ratio of reactant/product concentrations, adjusted for reaction orders: 6.01865
Formula used: [B] / [A]
2. Ratio of forward/reverse reaction rates: 6
Discrepancy between the two values: 0.3108 %
Reaction IS in equilibrium (within 1% tolerance)
True
# Register the new chemical ("S")
chem_data.add_chemical("S")
# Add the reaction A <-> S (fast shunt, poor thermodynical energetic advantage)
chem_data.add_reaction(reactants=["A"], products=["S"],
forward_rate=150., reverse_rate=100.)
chem_data.describe_reactions()
Number of reactions: 2 (at temp. 25 C)
0: A <-> B (kF = 30 / kR = 5 / delta_G = -4,441.7 / K = 6) | 1st order in all reactants & products
1: A <-> S (kF = 150 / kR = 100 / delta_G = -1,005.1 / K = 1.5) | 1st order in all reactants & products
Set of chemicals involved in the above reactions: {'S', 'A', 'B'}
# Send a plot of the network of reactions to the HTML log file
chem_data.plot_reaction_network("vue_cytoscape_2")
[GRAPHIC ELEMENT SENT TO LOG FILE `down_regulate_1.log.htm`]
dynamics = UniformCompartment(chem_data=chem_data, preset="mid") # Notice we're over-writing the earlier "dynamics" object
dynamics.set_conc(conc={"A": 50.}, snapshot=True)
dynamics.describe_state()
SYSTEM STATE at Time t = 0:
3 species:
Species 0 (A). Conc: 50.0
Species 1 (B). Conc: 0.0
Species 2 (S). Conc: 0.0
Set of chemicals involved in reactions: {'S', 'A', 'B'}
Run the reaction¶
dynamics.set_diagnostics() # To save diagnostic information about the call to single_compartment_react()
# The changes of concentrations vary very rapidly early on; automated variable timesteps will take care of that
dynamics.single_compartment_react(initial_step=0.001, duration=0.3,
snapshots={"initial_caption": "1st reaction step",
"final_caption": "last reaction step"},
variable_steps=True)
57 total step(s) taken
Number of step re-do's because of negative concentrations: 0
Number of step re-do's because of elective soft aborts: 2
Norm usage: {'norm_A': 38, 'norm_B': 37, 'norm_C': 36, 'norm_D': 36}
dynamics.plot_history(colors=["darkturquoise", "green", "red"],
title="Coupled reactions A <-> B and A <-> S (fast but disadvantaged energetically)",
show_intervals=True)
# Verify that all the reactions have reached equilibrium
dynamics.is_in_equilibrium()
0: A <-> B
Final concentrations: [A] = 5.887 ; [B] = 35.24
1. Ratio of reactant/product concentrations, adjusted for reaction orders: 5.98644
Formula used: [B] / [A]
2. Ratio of forward/reverse reaction rates: 6
Discrepancy between the two values: 0.226 %
Reaction IS in equilibrium (within 1% tolerance)
1: A <-> S
Final concentrations: [A] = 5.887 ; [S] = 8.873
1. Ratio of reactant/product concentrations, adjusted for reaction orders: 1.50724
Formula used: [S] / [A]
2. Ratio of forward/reverse reaction rates: 1.5
Discrepancy between the two values: 0.4824 %
Reaction IS in equilibrium (within 1% tolerance)
True
# Specify the chemicals (notice that we're starting with new objects)
new_chem_data = chem(names=["A", "B", "S"])
# Reaction A <-> B (as before)
new_chem_data.add_reaction(reactants=["A"], products=["B"],
forward_rate=30., reverse_rate=5.)
# Reaction A <-> S (slow shunt, excellent thermodynamical energetic advantage)
new_chem_data.add_reaction(reactants=["A"], products=["S"],
forward_rate=3., reverse_rate=0.1)
new_chem_data.describe_reactions()
Number of reactions: 2 (at temp. 25 C)
0: A <-> B (kF = 30 / kR = 5 / delta_G = -4,441.7 / K = 6) | 1st order in all reactants & products
1: A <-> S (kF = 3 / kR = 0.1 / delta_G = -8,431.4 / K = 30) | 1st order in all reactants & products
Set of chemicals involved in the above reactions: {'S', 'A', 'B'}
dynamics = UniformCompartment(chem_data=new_chem_data, preset="small_rel_change")
dynamics.set_conc(conc={"A": 50.}, snapshot=True)
dynamics.describe_state()
SYSTEM STATE at Time t = 0:
3 species:
Species 0 (A). Conc: 50.0
Species 1 (B). Conc: 0.0
Species 2 (S). Conc: 0.0
Set of chemicals involved in reactions: {'S', 'A', 'B'}
Run the reaction¶
dynamics.set_diagnostics() # To save diagnostic information about the call to single_compartment_react()
# The changes of concentrations vary very rapidly early on; automated variable timesteps will take care of that
dynamics.single_compartment_react(initial_step=0.005, duration=7.0,
snapshots={"initial_caption": "1st reaction step",
"final_caption": "last reaction step"},
variable_steps=True)
454 total step(s) taken
Number of step re-do's because of negative concentrations: 0
Number of step re-do's because of elective soft aborts: 1
Norm usage: {'norm_A': 17, 'norm_B': 17, 'norm_C': 16, 'norm_D': 16}
dynamics.plot_history(colors=["darkturquoise", "green", "red"],
title="Coupled reactions A <-> B and A <-> S (slow but with energetic advantage)")
dynamics.explain_time_advance()
From time 0 to 0.0025, in 2 steps of 0.00125 From time 0.0025 to 0.03281, in 97 steps of 0.000313 From time 0.03281 to 0.05859, in 55 steps of 0.000469 From time 0.05859 to 0.07617, in 25 steps of 0.000703 From time 0.07617 to 0.09199, in 15 steps of 0.00105 From time 0.09199 to 0.1062, in 9 steps of 0.00158 From time 0.1062 to 0.1205, in 6 steps of 0.00237 From time 0.1205 to 0.2237, in 29 steps of 0.00356 From time 0.2237 to 0.3999, in 33 steps of 0.00534 From time 0.3999 to 0.6402, in 30 steps of 0.00801 From time 0.6402 to 0.9525, in 26 steps of 0.012 From time 0.9525 to 1.349, in 22 steps of 0.018 From time 1.349 to 1.998, in 24 steps of 0.027 From time 1.998 to 3.66, in 41 steps of 0.0405 From time 3.66 to 4.876, in 20 steps of 0.0608 From time 4.876 to 5.971, in 12 steps of 0.0912 From time 5.971 to 6.929, in 7 steps of 0.137 From time 6.929 to 7.134, in 1 step of 0.205 (454 steps total)
Check the final equilibrium¶
# Verify that all the reactions are close to equilibrium
dynamics.is_in_equilibrium(tolerance=12)
0: A <-> B
Final concentrations: [A] = 1.488 ; [B] = 9.019
1. Ratio of reactant/product concentrations, adjusted for reaction orders: 6.06134
Formula used: [B] / [A]
2. Ratio of forward/reverse reaction rates: 6
Discrepancy between the two values: 1.022 %
Reaction IS in equilibrium (within 12% tolerance)
1: A <-> S
Final concentrations: [A] = 1.488 ; [S] = 39.49
1. Ratio of reactant/product concentrations, adjusted for reaction orders: 26.5412
Formula used: [S] / [A]
2. Ratio of forward/reverse reaction rates: 30
Discrepancy between the two values: 11.53 %
Reaction IS in equilibrium (within 12% tolerance)
True
Please note the much-longer timescale from the earlier plots¶
If we look at early time interval, this is what it looks like:
dynamics.plot_history(colors=["darkturquoise", "green", "red"],
title="Same plot as above, both only showing initial detail", xrange=[0, 0.3])
# Look at where the curves intersect
dynamics.curve_intersect("A", "B", t_start=0, t_end=0.1)
(0.02405919499545674, 23.73396682504195)
dynamics.curve_intersect("A", "S", t_start=0.1, t_end=0.2)
(0.14412951669101942, 6.026379520544665)